R&D
Insecticidal and synergistic effects of Majoranahortensis essential oil and some of its major constituents

R&D

Author: Mostafa A.Abbassy1, Smir A.M.Abdelgaleil2, Rasha Y.A.Rabie1
1Department of Pest Control and Environmental Protection, Faculty of Agriculture (Damanhour), Alexandria University, Damanhour, Egypt
2Department of Pesticide Chemistry, Faculty of Agriculture, Alexandria University, El-Shatby, Alexandria, Egypt
The essential oil from leaves of Majorana hortensis Moench (Lamiaceae) was isolated by hydrodistillation with a yield of 1.6% (wt ⁄ wt). The insecticidal activity of the oil was evaluated against fourth instars of Spodoptera littoralis Boisduval (Lepidoptera: Noctuidae) and adults of Aphis fabae L. (Hemiptera: Aphididae). The oil showed a remarkable toxic effect against S. littoralis in a topical application assay (LD50 = 2.48 lg per larva) and in a residual film assay (LC50 = 3.14 g ⁄ l). The oil of M. hortensis also exhibited a pronounced toxic effect against A. fabae adults with LC50 values of 1.86 and 2.27 g ⁄ l in rapid dipping and residual film assays, respectively. Gas chromatography-mass spectrometry analyses of M. hortensis essential oils revealed the presence of 31 compounds and the main components were terpinen-4-ol (30.0%), γ-terpinene (11.3%), and trans-sabinene hydrate (10.8%). Repeated column chromatography of M. hortensis oil on silica gel led to the isolation of two major constituents, which were characterized based on 1H-nuclear magnetic resonance and mass spectrometric data, as terpinen-4-ol and γ-terpinene. These two components were examined for their insecticidal and synergistic activities towards S. littoralis and A. fabae. Terpinen-4-ol andγ-terpinene exhibited a significant insecticidal activity against both insects, butγ-terpinene was more toxic than terpinen-4-ol. When tested in a binary mixture with the synthetic insecticides profenofos and methomyl, it was found that both compounds enhanced the insecticidal activity of these insecticides by two- to threefold. These results show that terpinen-4-ol andγ-terpinene have a synergistic effect on the insecticidal activities of synthetic insecticides profenofos and methomyl.
chemical composition, terpinen-4-ol, γ-terpinene, Lamiaceae, Spodoptera littoralis, Aphis fabae, Hemiptera, Aphididae, Lepidoptera, Noctuidae
Adverse environmental effects and the development of insect resistances to synthetic insecticides have created the need for degradable and target-specific insecticides. Therefore, an effort is warranted to find alternatives to currently used insecticides. One source of potential new insecticides is natural products synthesized by plant species. Many plant products and chemicals have arvicidal, pupicidal, and adulticidal activities, most being repellants, oviposition deterrents, or antifeedants against both agricultural pests and medically important insect species (Thorsell et al., 1979). Not only might certain natural products be a source of new insecticides, but botanical derivatives may also be more environmentally benign than synthetic chemicals.
Essential oils are a complex mixture of mainly monoterpenes, sesquiterpenes, and their oxygenated derivatives, as well as phenylpropenes and specific sulphur- or nitrogen-containing substances (Cowan, 1999). In addition to their antibacterial and antifungal activities, many essential oils isolated from plant species have been found to act against various insects (Abbassy, 1981; Curtis et al., 1991; Regnault-Roger, 1997).
Marjoram, Majorana hortensis Moench (Lamiaceae), is an herbaceous, perennial plant, native to the Mediterranean basin. It is cultivated not only in Mediterranean countries, but also in Central and Eastern Europe (Novak et al., 2000). Marjoram is mainly used as a spice in the food industry (Burdock, 1995). The plant is also medicinally valuable because of its stimulant and antispasmodic properties, and it is a good general tonic, treating various disorders of the digestive and respiratory systems (Chevallier, 1996).
In Egypt, the cotton leafworm, Spodoptera littoralis Boisduval (Lepidoptera: Noctuidae), and the bean aphid, Aphis fabae L. (Hemiptera: Aphididae), are considered two of the most destructive insect pests of a variety of crops (Abdelgaleil et al., 2002; Ahmed et al., 2007). It has been reported that M. hortensis oil acts against the larvae and eggs of Aedes aegypti L. (Salehet al., 1983), but insecticidal activity against S. littoralis and A. fabae has not been studied.
As part of the research program investigating bioactive compounds from Egyptian plants as insect control agents, in this study the hydrodistillated essential oil of M. hortensis was tested for its insecticidal activity against S. littoralis and A. fabae. The chemical composition of the oil was examined by gas chromotography-mass spectrometry (GC-MS). Terpinen-4-ol and γ-terpinene, the major constituents of the oil, were isolated and their insecticidal and toxic effects combined with synthetic insecticides were also studied.
Wild marjoram was collected from Sinai Peninsula desert (27°55¢N, 34°21¢E), Egypt, in September 2007. The plant material was identified by Prof. Fath Allah Zitoon of Alexandria University. A voucher specimen (FA-MH-07 ⁄ 7) has been deposited in the herbarium of the Faculty of Agriculture, Alexandria University.
A susceptible strain of S. littoralis was obtained from the Bioassay Laboratory, Faculty of Agriculture, Alexandria University. The colony was reared under laboratory conditions on caster bean leaves, Ricinus communis L. (Euphorbiaceae), at 26 ± 2°C and 70 ± 5% r.h. (Eldefrawi et al., 1964). A field strain of A. fabae was collected from a farm in Elbehera Governorate, north Egypt (31°6¢N, 30°19¢E), and reared under laboratory conditions on faba bean plants at 22–25°C and 65 ± 5% r.h. (Norman Sution, 1967).
Essential oil of air-dried leaves of M. hortensis was extracted by hydrodistillation in a Clevenger-type apparatus for 2 h (Guenther, 1952). The oil was dried over anhydrous sodium sulfate. The essential oil was diluted in diethyl ether and 1 ll was injected into a gas chromatograph (Trace GC 2000; Thermo, Cairo, Egypt)-mass spectrometer (SSQ 7000; Finnigan, Cairo, Egypt). The GC column was a DB-5 (5% phenylmethylpolysiloxane) capillary column (60 m×0.25 mm i.d.). The GC conditions were as follows: injector temperature, 220°C; column temperature, isothermal at 40 C for 2 min, then programmed to 250°C at 5°C⁄ 2 min and held isothermal for 2 min; ion source temperature, 200°C. Helium was used as the carrier gas at a rate of 1 ml ⁄ min. The effluent of the GC column was introduced directly into the ion source of the MS. Spectra were obtained in the EI mode with 70 eV ionization energy. The sector mass analyzer was set to scan from 40 to 400 amu for 5 s.
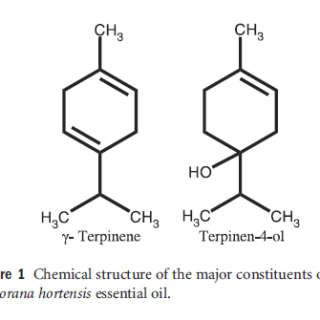
Figure1 Chemicals structure of the major constituents of Majorana hortensis essential oil
The essential oil of M. hortensis (10 g) was hromatographed on silica gel (250 g) using a hexane ⁄ acetone solvent system, starting with fractions of 100 ml of hexane (10×), 2% acetone ⁄ hexane (20×), 5% acetone ⁄ hexane (10×), 10% acetone ⁄ hexane (5×), and finally acetone (5×). The resulting fractions were pooled into two main fractions based on their thin-layer chromatography (TLC) profiles. The first main fraction (fractions 20–26, 4.1 g) was further purified on a silica gel column eluted with 2% acetone ⁄ hexane to give 2.3 g of γ-terpinene. Similarly, the second main fraction (fractions 38–40, 8.1 g) was subjected to a silica gel column eluted with 10% acetone ⁄ hexane to offer 4.3 g of terpinen-4-ol. The structures of γ-terpinene and terpinen-4-ol(Figure 1) were determined spectroscopically by 1H-nuclear magnetic resonance (NMR) (JEOL JNM ECD 500 Spectrometer; JEOL Co., Tokyo, Japan) and MS (JEOL JMS-AX500 Mass Spectrometer).
Topical application assay. Fourth instars of S. littoralis were used to assess the larvicidal activity of the M. hortensis oil and its major isolated compounds γ-terpinene and terpinen-4-ol. Serial concentrations of the oil, the isolated compounds, and a reference insecticide, profenofos, were prepared in acetone. One microliter of test solution was applied on the dorsum of larvae by a microapplicator. The oil was tested at concentrations of 1.0, 1.5, 2.5, 3.5, 4.5, and 6.0 lg per larva.γ-Terpinene and terpinen-4-ol were tested at concentrations of 5, 10, 20, 30, 40, and 50 lg per larva. Profenofos was tested at concentrations of 0.1, 0.2, 0.3, 0.5, 0.7, and 1.0 lg per larva. Three replicates of 10 larvae were used for each dose and control treatments. The treated larvae were then transferred to glass cups and supplied with fresh castor bean leaves. Mortality (%) was recorded at 24 h posttreatment. The lethal doses causing 50% mortality (LD50) expressed as microgram per larva were calculated from log-dose mortality regression lines (Finney, 1971).
Residual film assay. To evaluate their insecticidal activities against S. littoralis and A. fabae, solutions of M.hortensis oil and the isolated compounds c-erpinene and terpinen-4-ol were prepared first in acetone. Serial dilutions of these solutions were prepared with distilled water containing 0.05% of a wetting agent (Triton-X100) to give a series of concentrations (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, and 6.0 g ⁄ l). In the case of S.littoralis, castor bean leaf disks (20 mm in diameter) were immersed in test solutions for 10 s and left to dry. Control disks were immersed in the same solution without the oil and pure compounds. Ten treated disks were transferred into a glass cup with 10 fourth instars. Three replicates of each concentration and control were used. Profenofos was used as a reference insecticide and tested under the same conditions. For A. fabae a similar protocol was followed except for using faba bean disks. The treated disks were placed in glass cylinders with gauze lids containing 10 aphids. Methomyl was used as the recommended insecticide for bean aphids. Mortality (%) was recorded after 24 h. LC50 values were calculated from log-dose mortality regression lines.
Rapid dipping assay. The M. hortensis oil and the isolated compounds γ-terpinene and terpinen-4-ol were tested at a series of concentrations (0.5, 1.5, 2.0, 3.0,and 4.0 g ⁄ l). Ten aphids were placed in a short glass cylinder with the bottom covered with gauze and internally coated with fluon. The aphids were immersed by placing the cylinder in a shallow Petridish containing 2 ml of test compound solution for 10 s. Mortality (%) was recorded after 24 h and LC50 was calculated from log-dose mortality regression lines.
Joint toxic effect of isolated compounds and insecticides. This assay was carried out to examine the joint toxic effect of the isolated compounds, γ-terpinene and terpinen-4-ol, and the synthetic insecticides profenofos and methomyl on S. littoralis and A. fabae, respectively. Mortality percentages corresponding to the LD25 and ⁄ or LC25 of the isolated compounds and the insecticides were determined from regression lines. The summation of mortality percentages for each isolated compound and insecticide is considered as expected mortality. A binary mixture of isolated compound with insecticide was prepared at a ratio of LD25:LD25 (1:1) and ⁄ or LC25:LC25 (1:1). The resulting mixtures were tested against the insects. Mortality (%) caused by these mixtures was recorded after 24 h of treatment and considered as observed mortality. A co-toxicity factor was taken as a criterion for evaluation of the joint toxic effect as follows:
co-toxicity factor = [(OM − EM)/EM]×100;
where OM is the observed mortality (%) and EM is the expected mortality (%). A positive factor of +20 or higher means potentiation, a negative factor of )20 or lower means antagonism, and values between )20 and +20 imply an additive effect (Mansour et al., 1966; Abbassy et al., 1979).
For precise estimation of the synergistic activity of the isolated compounds on insecticides, the LD50 and ⁄ or LC50 values of the mixtures of each isolated compound with an insecticide were determined by treating the insects with a series of concentrations of the mixture at a ratio of 1:1. The degree of interaction was estimated as synergism ratio (SR) as described by Chadwick (1961) and Metcalf (1967) as follows:
SR = LD50 or LC50 of pesticide alone
/LD50 or LC50 of mixture.
Thus, a synergism ratio of 1.0 indicates that the toxicity of the pesticide alone is equal to the toxicity of the mixture of pesticide and isolated compound. A lower value indicates antagonism, a higher value synergism.
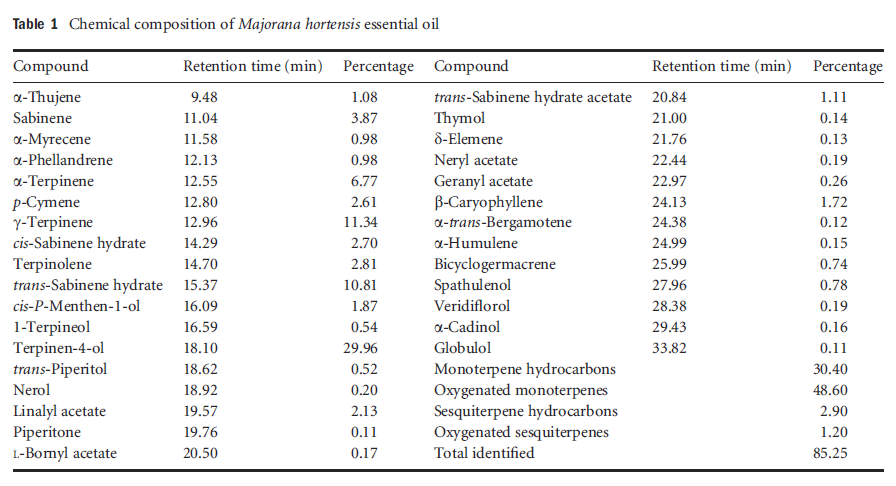
Table1 Chemical composition of Majorana hortensis essential oil
Mortality of each dose and ⁄ or concentration was calculated after 24 h of treatment as a mean of three replicates with 10 insects each. The mortality data were subjected to Probit analysis (Finney, 1971) to obtain the LD50 and LC50 values, using SPSS 12.0 (SPSS, Chicago, IL, USA). The values of LD50 and LC50 were considered significantly different, if the 95% confidence limits did not overlap.
Hydrodistillation of M. hortensis leaves produced a white oil with a yield of 1.6% (wt ⁄ wt) on a dry-weight basis. Thirty-one components representing 85.3% of the oil were identified by GC-MS (Table 1). The major components were terpinen-4-ol (30.0%), γ-terpinene (11.3%), and trans-sabinene hydrate (10.8%).
The oil showed a remarkable insecticidal activity against S. littoralis with LD50 = 2.48 lg per larva and LC50 = 3.14 g ⁄ l in the topical application and residual film assay, respectively (Table 2). Similarly, the oil exhibited a potent activity against A. fabae. The oil was significantly more effective against A. fabae when tested by the rapid dipping assay (LC50 = 1.86 g ⁄ l) than by the residual film assay (LC50 = 2.27 g ⁄ l).
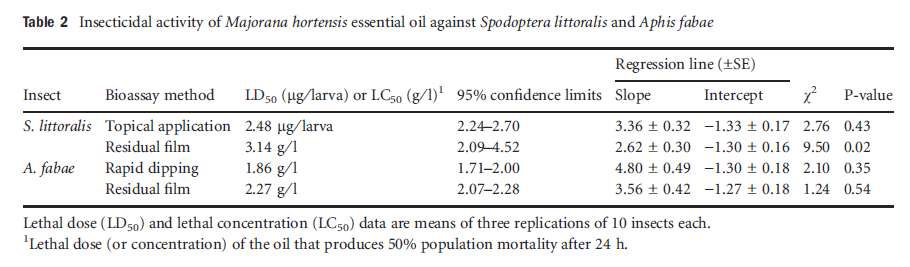
Table2 Insecticidal activity of Majorana hortensis essential oil against Spodoptera littoralis and Aphis fabae
In both the topical application and the residual film assays with fourth instars of S. littoralis, the LD50 and LC50 values for γ-terpinene (LD50 = 11.86 lg per larva and LC50 = 23.9 g ⁄ l) were lower than for terpinen-4-ol (LD50 = 16.20 lg per larva and LC50 = 32.9 g ⁄ l), but the 95% confidence intervals overlapped, so there was no significant difference in insecticidal effect between the two compounds (Table 3). Both natural compounds showed a pronounced insecticidal activity, but they were significantly less effective than profenofos. In the rapid dipping and the residual assay, the toxicity of γ-terpinene and terpinen-4-ol against A. fabae adults was not significantly different (LC50 = 12.2 and 14.9 g ⁄ l, respectively; Table 4). The two isolated compounds and the insecticide methomyl were more toxic when tested by the rapid dipping assay than by the residual film assay. In the residual film assay the isolated compounds were more effective against A. fabae than against S. littoralis.
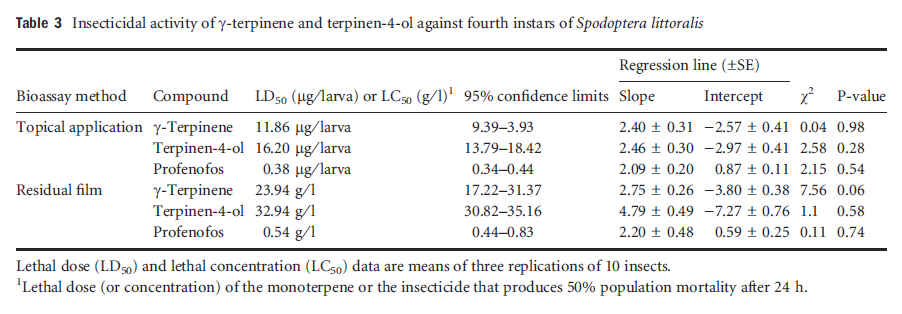
Table3 Insecticidal activity of gamma-terpinene and terpinene-4-ol against fourth instars of Spodoptera littoralis
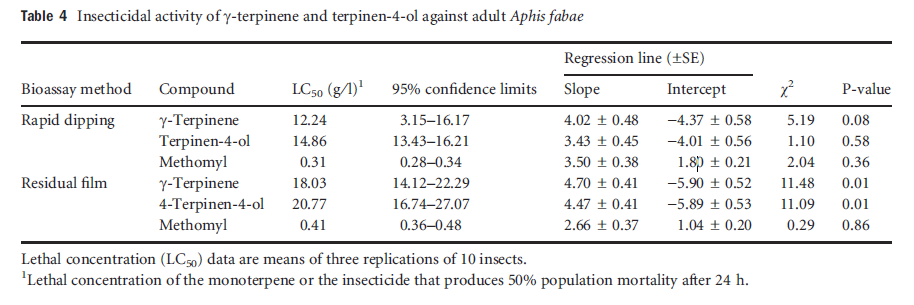
Table4 Insecticidal activity of gamma-terpinene and terpinene-4-ol against adult Aphis fabae
The isolated compounds γ-terpinene and terpinen-4-ol enhanced the toxicity of profenofos against S. littoralis with co-toxicity factors of +35.3 and +26.8, respectively. In addition, the isolated compounds also increased the toxicity of methomyl towards the adults of A. fabae (Table 5). As co-toxicity factor values exceeded +20, the isolated compounds caused potentiation of toxicity of insecticides.
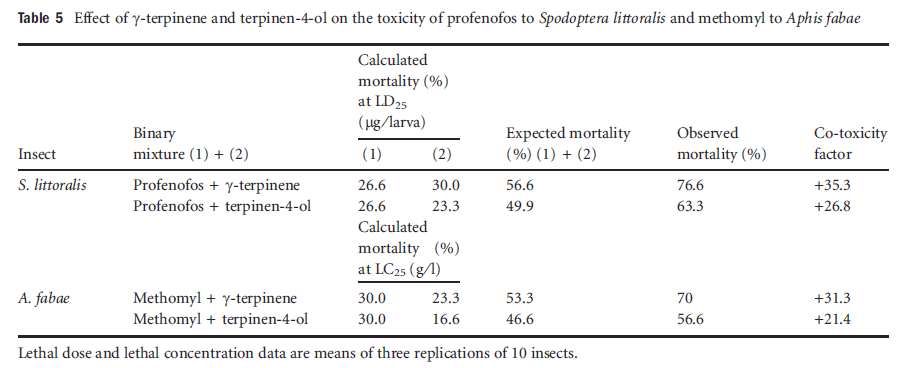
Table5 Effect of gamma-terpinene and terpinen-4-ol on the toxicity of profenofos to Spodoptera littoralis and methomyl to Aphis fabae
The enhancement effect of the isolated compounds on toxicity of pesticides against the tested insects was confirmed by the synergism ratio. γ-terpinene and terpinen-4-ol increased the toxicity of profenofos against S. littoralis by twofold, and increased the toxicity of methomyl against A. fabae by threefold (Table 6).
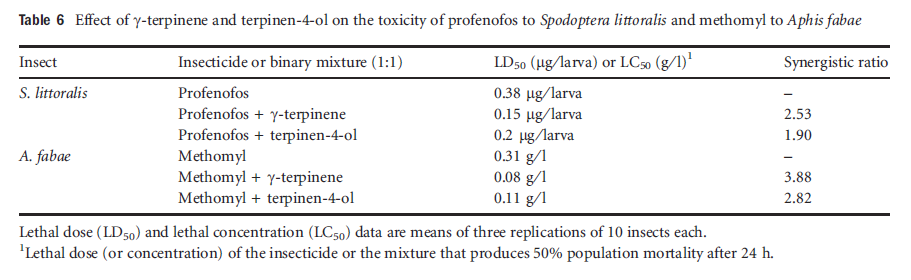
Table6 Effect of gamma-terpinene and terpinen-4-ol on the toxicity of profenofos to Spodoptera littoralis and methomyl to Aphis fabae
Oxygenated monoterpenes and monoterpene hydrocarbons were the major groups of compounds in M. hortensis oil, as shown by GC-MS analysis. Sesquiterpene hydrocarbons and oxygenated sesquiterpenes were also present as minor constituents. The major constituents of the oil, terpinen-4-ol, γ-terpinene, and trans-sabinene hydrate, were similar to those of the oils of other M. hortensis plants growing in Egypt (El-Ghorab et al., 2004), or Russia, Germany, France, Hungary, Romania, Portugal, and Turkey (Misharina et al., 2003), although concentrations of the major compounds and the remaining chemical profile of the oil could differ probably due to both environmental and genetic factors.
The strong insecticidal activity of M. hortensis oil encouraged us to isolate its major components to know the compounds responsible for the activity observed. Two compounds were isolated and identified as γ-terpinene and terpinen-4-ol. Both compounds possessed a pronounced insecticidal activity against S. littoralis and A. fabae. However, the two compounds were less active than trans-ethyl cinnamate and piperitone, two compounds that were isolated from the essential oil of Artemisia judaica L. (Abdelgaleil et al., 2008) and the glucosides simmondsin and simmondsin 2-ferulate, isolated from Simmondsia chinensis (Link) Schneider (Abbassy et al., 2007), tested by a topical application assay against the third instar of S. littoralis. It has been reported that γ-terpinene and terpinen-4-ol have insecticidal activity against the Colorado potato beetle, Leptinotarsa decemlineata Say (Kordali et al., 2007). Likewise, terpinen-4-ol showed toxic effects on the stored-product beetles Sitophilus granarius L. and Sitophilus oryzae L. (Lee et al., 2001; Kordali et al., 2006). γ-terpinene showed larvicidal activity against the yellow fever mosquito Aedes aegypti L. and Aedes albopictus Skuse (Cheng et al., 2008).
Comparing the activity of M. hortensis oil with that of γ-terpinene and terpinen-4-ol revealed that the oil was more toxic against S. littoralis and A. fabae than the isolated compounds. This finding suggests that the complexity of the essential oil led to a complex activity probably with synergistic and ⁄ or antagonistic phenomena between allelochemicals as reported by Regnault-Roger (1997) for other essential oils. A similar conclusion has been drawn for the essential oils of some Ocimum spp. against post-harvest pests (Bekele Hassanali, 2001) and the essential oils of Conyza newii Oliv. Hiern. and Plectranthus marruboides Benth. against Anopheles gambiae Giles (Omolo et al., 2005). The mechanism underlying the nsecticidal activity of the M. hortensis oil, γ-terpinene, and terpinen-4-ol is unknown, but some essential oils and monoterpenes are known to act as acetylcholinesterase inhibitors, and others as neurotoxic agents, such as antagonists of octopamine receptors (Isman, 2000).
Besides their individual insecticidal activity against S. littoralis and A. fabae, the isolated compounds were tested in binary blends with the insecticides profenofos and methomyl. The results indicated that the isolated compounds enhanced the effectiveness of the insecticides against the tested insects two- to threefold. This synergistic effect may lead to the reduction of the application rates of these insecticides which would be less harmful to the environment. Although the combined toxic effects of insecticide ⁄ insecticide ixtures and ⁄ or insecticide ⁄ insect growth regulator mixtures on S. littoralis are well investigated (Mansour et al., 1966; Abbassy et al., 1979; El-Guindy et al., 1983), there have been no earlier studies on the combined action of monoterpene ⁄ insecticide mixtures against this insect.
In conclusion, our findings indicate that the oil of M. hortensis and its major constituents, γ-terpinene and terpinen-4-ol, should be explored as potential natural insecticides against S. littoralis and A. fabae. The synergistic effect of γ-terpinene and terpinen-4-ol with insecticides could help to decrease the negative effects of synthetic chemicals such as residues in products, development of insect resistance, and environmental pollution.
> Abbassy MA (1981) Naturally occurring chemicals for pest control. V. Insecticidal and synergistic citrus oils isolated from certain food and medicinal plants. Proceedings of the Fourth Arab Pesticide Conference, (ed. by AS El-Nawawy), Vol. IIIA. pp. 409–414. Tanta University, Tanta, Egypt.
>Abbassy MA, Hosny A, Lamie OM & Choukri O (1979) Insecticidal and synergistic citrus oils isolated from Citrus peels. Mededelingen van de Faculteit Landbouwwetenschappen der Rijksuniversiteit Gent 44: 21–29.
> Abbassy MA, Abdelgaleil SAM, Belal AH & Abdel Rasoul MAA (2007) Insecticidal, antifeedant and antifungal activities of two glucosides isolated from the seeds of Simmondsia chinensis (link). Industrial Crops and Products 26: 345–350.
> Abdelgaleil SAM, El-Aswad AF & Nakatani M (2002) Molluscicidal and anti-feedant activities of diterpenes from Euphorbia paralias L. Pest Management Science 58: 479–482.
> Abdelgaleil SAM, Abbassy MA, Belal AH & Abdel Rasoul MAA (2008) Bioactivity of two monoterpenoids isolated from Artemisia judaica L. Bioresouce Technology 99: 5947–5950.
> Ahmed AAI, Gesraha MA & Zebitz CPW (2007) Bioactivity of two neem products on Aphis fabae. Journal of Applied Science Research 3: 392–398.
> Bekele J & Hassanali A (2001) Blend effects in the toxicity of the essential oil constituents of Ocimum kilimandscharicum and Ocimum kenyense (Labiateae) on two post-harvest insect pests. Phytochemistry 57: 385–391.
> Burdock GA (1995) Fenaroli’s Handbook of Flavor Ingredients. Vol. I: Natural Flavors. CRC Press, Boca Raton, FL, USA.
> Chadwick PR (1961) A comparison of Safraxon and piperonyl butoxide as pyrethrum synergists. Pyrethrum Post 6: 60–73.
> Cheng SS, Chua M-T, Chang EH, Huang C-G, Chen W-J &
> Chang S-T (2008) Variations in insecticidal activity and chemical compositions of leaf essential oils from Cryptomeria japonica at different ages. Bioresource Technology 100: 465–470.
> Chevallier A (1996) The Encyclopedia of Medicinal Plants. Dorling Kindersley, London, UK.
> Cowan MM (1999) Plant products as antimicrobial agents.
> Clinical Microbiology Reviews 12: 564–582.
> Curtis CF, Lines JD, Lu B & Renz A (1991) Natural and synthetic repellents. Control of Disease Vectors in the Community (ed. by CF Curtis), pp. 75–92. Wolfe Publishing, London, UK.
> Eldefrawi ME, Toppozada A, Mansour N & Zeid M (1964)
> Toxicological studies on the Egyptian cotton leafworm Prodenia litura L. susceptibility of different larval instars of Prodenia to insecticides. Journal of Economic Entomology 57: 591–593.
> El-Ghorab AH, Mansour AF & El-massry KF (2004) Effect of extraction methods on the chemical composition and antioxidant activity of Egyptian marjoram (Majorana hortensis Moench). Flavour and Fragrance Journal 19: 54–61.
> El-Guindy MG, El-Refaia AM & Abdel-Sattar MM (1983) The joint action of mixtures of insecticides, or of insect growth regulators and insecticides, on susceptible and diflubenzuron-resistant strains of Spodoptera littoralis Boisd. Pesticide Science 14: 246–252.
> Finney DJ (1971) Probit Analysis, 3rd edn. Cambridge University Press, Cambridge, UK.
> Guenther E (1952) The Essential Oils Vol. 4: Individual essental oils of the plant families Rutaceae and Labiatae (ed. by Van Nostrand Co.), p. 81. New York, USA.
> Isman MB (2000) Plant essential oils for pest and disease management. Crop Protection 19: 603–608.
> Kordali S, Aslan I, Calmasur O & Cakir A (2006) Toxicity of essential oils isolated from three Artemisia species and some of their major components to granary weevil, Sitophilus granarius (L.) (Coleoptera: Curculionidae). Industrial Crops and Products 23: 162–170.
> Kordali S, Kesdek M & Cakir A (2007) Toxicity of monoterpenes against larvae and adults of Colorado potato beetle, Leptinotarsa decemlineata Say (Coleoptera: Chrysomelidae). Industrial Crops and Products 26: 278–297.
> Lee BH, Choi WS, Lee SE & Park BS (2001) Fumigant toxicity of essential oils and their constituent compounds towards the rice weevil, Sitophilus oryzae (L.). Crop Protection 20: 317–320.
> Mansour N, Eldefrawi ME, Toppozada A & Zeid M (1966) Toxicological studies on the Egyptian cotton leafworm, Prodenia litura. VI. Potentiation and antagonism of organophosphours and carbamate insecticides. Journal of Economic Entomology 59: 307–311.
> Metcalf RL (1967) Mode of action of insecticide synergists. Annual Review of Entomology 12: 229–256.
> Misharina TA, Polshkov AN, Ruchkina EL & Medvedeva IB
(2003) Changes in the composition of the essential oil of marjoram during storage. Applied Biochemistry and Microbiology 39: 311–316.
> Norman PA & Sution RA (1967) Host plants for melin aphid. Journal of Economic Entomology 60: 1205–1207.
> Novak J, Bitsch C, Langbehn J, Pank F, Skoula M et al. (2000) Ratios of cis- and trans-sabinene hydrate in Origanum majorana L. and Origanum microphyllum (Bentham) Vogel. Biochemical Systematics and Ecology 28: 697–704.
> Omolo MO, Okinyoa D, Ndiege IO, Lwande W & Hassanali A (2005) Fumigant toxicity of the essential oils of some African plants against Anopheles gambiae sensu stricto. Phytomedicine 12: 241–246.
> Regnault-Roger C (1997) The potential of botanical essential oils for insect pest control. Integrated Pest Management Review 2: 15–34.
> Saleh RS, Saleh MS & Gaboub IA (1983) Management of Aedes aegypti by some plant volatile extracts. Proceedings of the International Conference on Environmental Hazards of Agrochemicals in Developing Counties (ed. by AH El-Sebae), pp. 1143–1152. Alexandria University, Alexandria, Egypt.
> Thorsell WA, Mikiver M & Malam E (1979) Plant extracts as protectants against disease causing insects. Entomologisk Tidskrift 100: 138–141.